GROUP KUHN / Associated DFG Project Ku 749/8-1
Translocation of infecting viral DNA across the bacterial host membrane
Bacteriophages adsorb to the outer surface of host cells and inject their DNA into the host cells without mechanical lesions. Upto now very little is known about the molecular details of these events in the different phage-host systems. Previous work with T7 has shown that the phage capsids contain internal proteins that are injected into the periplasm before the DNA is released. It has been postulated that the internal phage proteins assemble a DNA translocation machinery in the periplasm, called ejectosome, that catalyses the transfer of the DNA into the host cell. In T4 the contractile phage tail is pushed through the outer membrane and contacts the inner membrane by a still unknown mechanism. At present, it is unknown how the phage tail really contacts the host membrane and how the DNA is translocated.
In our project the phage-host interaction and DNA translocation through the host membrane is studied. For the podovirus T7 the internal proteins (gp14, gp15, gp16, gp6.7) have now been purified and are studied for complex formation, DNA binding and liposome binding. The assembly of the ejectosome complexes and the DNA translocation into the membrane vesicles will be studied (Fig. 1).
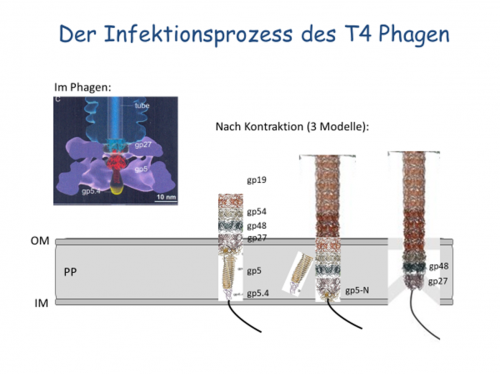
For the myovirus T4 the tail core partner candidates gp5, gp5.4, gp27, gp48 and gp54 will be studied for their capability to interact with the inner membrane (Fig. 2). Chemical crosslinking will be applied to identify inner membrane proteins that interact with the phage tail during the infection process. Partner proteins in the periplasm and the host membrane will be identified that specifically interact with the core tail tip. In addition, the fate of all tail core partner proteins in the periplasm during the infection process will be followed.
Principal Investigator(s)

Prof. Dr. Andreas Kuhn
University of Hohenheim
Institute of Biology (190) Molecular Microbiology
E-Mail: andreas.kuhn@uni-hohenheim.de
Homepage: FG Mikrobiologie (190h): Homepage Mikrobiologie (uni-hohenheim.de)

Dr. Dorothee Kiefer
University of Hohenheim
Institute of Biology (190) Molecular Microbiology
E-Mail: dorothee.kiefer@uni-hohenheim.de
Homepage: FG Mikrobiologie (190h): Homepage Mikrobiologie (uni-hohenheim.de)
PhD student(s)
Sabrina Wenzel, sabrina.wenzel@uni-hohenheim.de
Sandra Fischer, sandra_fischer@uni-hohenheim.de
Publications
- Lupo, D., S. Leptihn, G. Nagler, M. Haase, I. Molineux and A. Kuhn (2015). The T7 ejection nanomachine components gp15/gp16 form a spiral ring complex that binds DNA and a lipid membrane. Virology 486, 263-271.
- Wenzel, S., M.M. Shneider, P.G. Leiman, A. Kuhn and D. Kiefer (2020). The central spike complex of bacteriophage T4 contacts PpiD in the periplasm of Escherichia coli. Viruses 12, 1135.
